Biomedical Data Analysis · Translational Research · R&D Decision Support
Biopharma R&D, Translational Medicine & Publication-Ready Data Analysis
DeepTrans Bio helps biotech, CRO, hospital research, and academic teams turn biomedical data, literature evidence, and project questions into clear figures, structured reports, and decision-ready materials.
R&D Research Support
Project initiation, target review, competitive landscape
Translational Analytics
Biomarker screening, diagnostic models, clinical cohorts
Publication-Ready Analysis
Bioinformatics, SCI figures, statistical reports
Services
Three tracks aligned to the stages where data and analysis matter most.
Drug R&D Support
Research support for early-stage biopharma decision-making.
- Project initiation research
- Target / mechanism review
- Competitive landscape
- Decision report
Translational Medicine Analytics
Turn clinical and omics data into actionable evidence packages.
- Biomarker screening
- Diagnostic model analysis
- Clinical cohort statistics
- Evidence package
Academic & Publication-Ready Analysis
Bioinformatics and statistical analysis for manuscripts and revisions.
- Bioinformatics analysis
- SCI-ready figures
- Statistical reports
- Revision support
Who We Help
Teams that need biomedical data interpreted, visualized, and turned into decision materials.
Biotech & Pharma Teams
Early R&D, target selection, pipeline review, and competitive intelligence.
CRO / Service Providers
Bioinformatics support, figure generation, and report preparation for client deliverables.
Hospital & Translational Researchers
Clinical cohort analysis, biomarker discovery, and translational evidence packaging.
Academic Research Groups
Publication-ready analysis, manuscript revision support, and methodology review.
Featured Work
Representative analysis outputs. Figures generated from mock or de-identified data for layout demonstration.
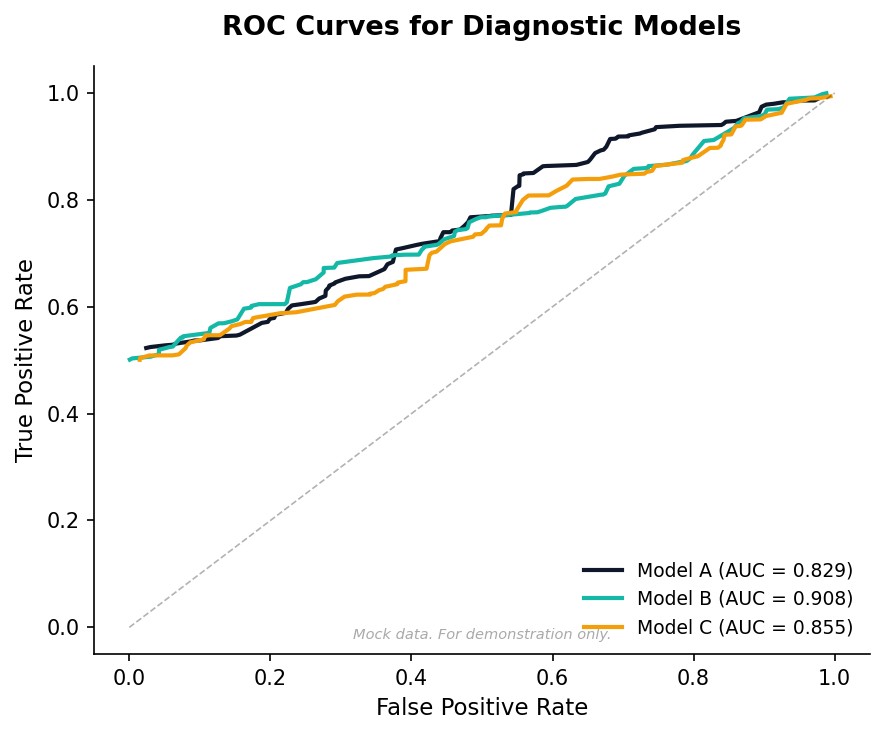
Biomarker Diagnostic Model
Screen candidate biomarkers and build a diagnostic scorecard with validation metrics.
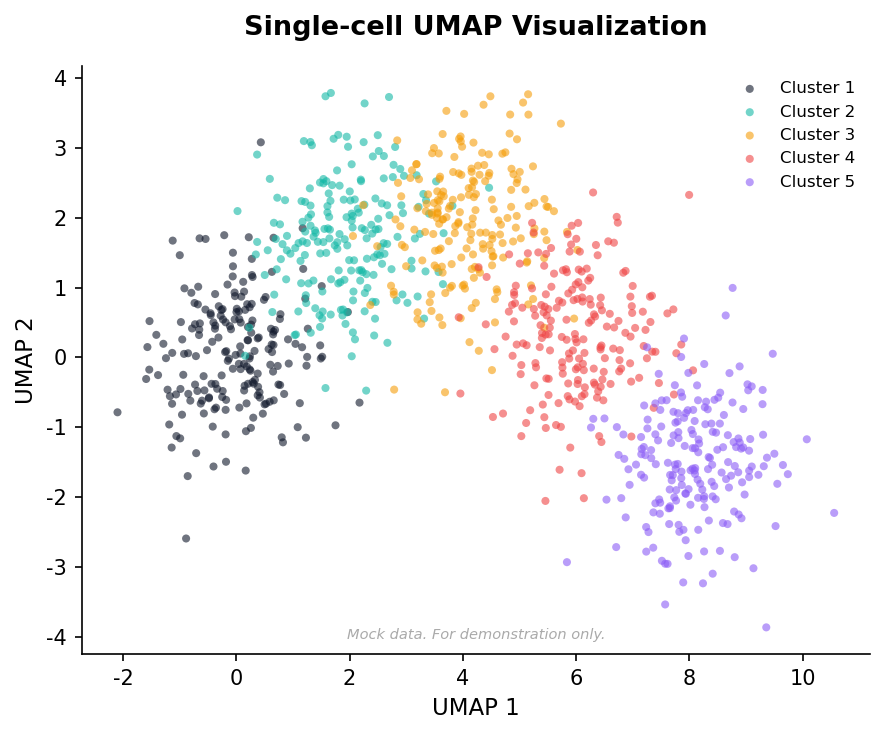
Single-Cell Transcriptomics
Characterize cell-type composition and marker expression from scRNA-seq data.
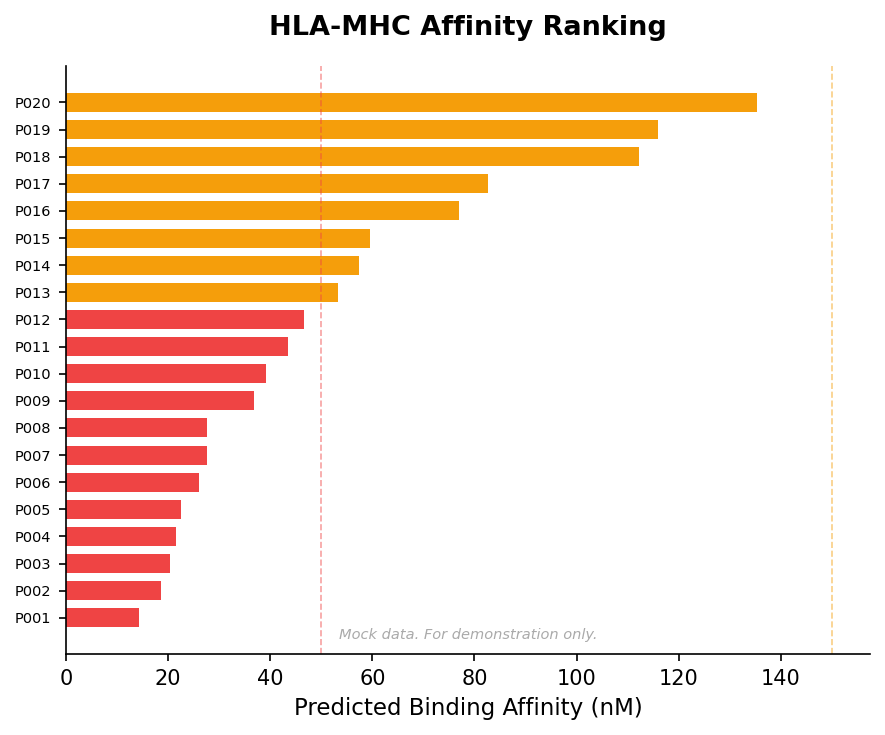
Neoantigen Prediction & Ranking
Rank tumor neoantigens by predicted immunogenicity and HLA binding affinity.
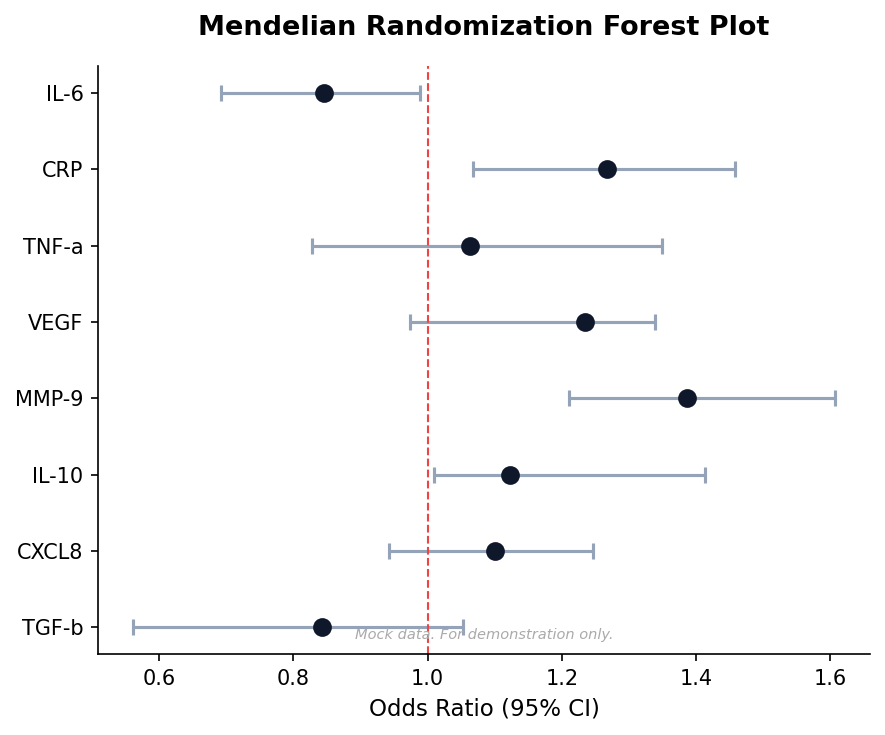
Mendelian Randomization
Infer causal relationships between exposures and outcomes using genetic instruments.
Why DeepTrans Bio
A background that spans wet-lab research, data analysis, and R&D project management.
Wet-lab + data-analysis background
Experience in molecular biology, immunology, and computational pipelines means analysis is grounded in biological context.
Biomedical R&D context understanding
Familiar with drug discovery timelines, translational milestones, and publication requirements.
Figure + report deliverables
Outputs are designed for internal review, grant applications, manuscripts, and stakeholder presentations.
Clear boundaries and auditable outputs
Every deliverable includes methods summary, data sources, and known limitations.
How It Works
A lightweight process designed for research teams with evolving questions.
Submit project context
Share your question, available data, and expected deliverables.
Receive scope & analysis plan
I assess fit, propose scope, timeline, and deliverables.
Get figures, reports, and recommendations
Iterative delivery with space for feedback and refinement.